  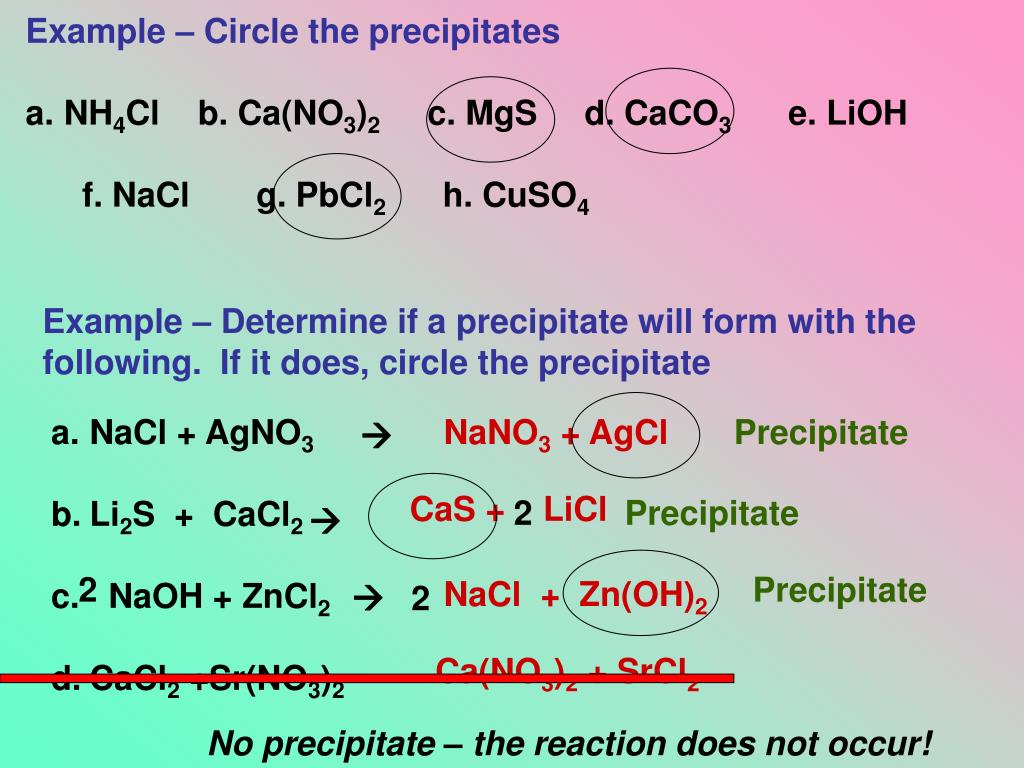
Once the precipitate has been recovered, the resulting powder may be called a "flower. A common sedimentation technique is centrifugation. Sedimentation refers to any procedure that separates the precipitate from the liquid portion of the solution, which is called the supernate. The products of the neutralization reaction will be water and calcium oxalate: H 2 C 2 O 4 (s) + Ca (OH) 2 (s) 2H 2 O () + CaC 2 O 4 (s) Because nothing is dissolved, there are no substances to separate into ions, so the net ionic equation is the equation of the three solids and one liquid. Additionally, these tests are also commonly used in chemistry labs. Equation Log Equation Cross Product Partial Derivative Implicit Derivative Tangent Complex. The lead would precipitate out as either PbCl2 or Pb(OH)2 and indicate that lead is present. If the particle size of the insoluble compound is very small or there is insufficient gravity to draw the solid to the bottom of the container, the precipitate may be evenly distributed throughout the liquid, forming a suspension. O 2 CO 2+ H 2 O SO 2+ H 2 S +2 H 2 O P 4+ O 2 P 2 O. B According to Table 4.2 'Guidelines for Predicting the Solubility of Ionic Compounds in Water', RbCl is soluble (rules 1 and 4), but Co(OH) 2 is not soluble (rule 5). The solid that is formed is called the precipitate. A chemical that causes a solid to form in a liquid solution is called a precipitant. Here's how it works: forming a solid from a solution is called precipitation. An example of a precipitation reaction is given below: CdSO4 ( aq) + K2S ( aq) CdS ( s) + K2SO4 ( aq) Both reactants are aqueous and one. The terminology can seem a bit confusing. This equation has the general form of an exchange reaction: AC + BD ADinsoluble + BC (12.4.2) (12.4.2) A C + B D A D i n s o l u b l e + B C. A double replacement reaction is specifically classified as a precipitation reaction when the chemical equation in question occurs in aqueous solution and one of the of the products formed is insoluble. Inorganic chemistry Precipitation in aqueous solution A common example of precipitation reaction in aqueous solution is that of silver chloride. They are used for purification, removing or recovering salts, for making pigments, and to identify substances in qualitative analysis. Precipitation reactions serve important functions.The solid that forms via a precipitation reaction is called the precipitate.AgNO 3 (aqueous) + KCl(aqueous) AgCl(precipitate) + KNO 3 (aqueous) In the above reaction, a white precipitate called silver chloride or AgCl is formed which is in the solid-state. The chemical equation for this precipitation reaction is provided below. To precipitate is to form an insoluble compound, either by decreasing the solubility of a compound or by reacting two salt solutions. This is the insoluble salt formed as a product of the precipitation reaction.In chemistry, precipitate is both a verb and a noun.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
